Latent viruses hide inside cells, remaining invisible to the immune system
Even in perfectly healthy people, viruses hide inside cells — sometimes for years and decades. They don’t cause symptoms, but they don’t disappear either: they simply lie dormant, evading the immune system. A new large-scale study has revealed which specific regions of the human genome determine how well the body keeps these hidden infections under control, and why in some people they remain harmless while in others they lead to serious diseases.
What are latent viruses
When we talk about a viral infection, we usually picture fever, coughing, and bed rest. But there is an entire class of viruses that work differently. They penetrate cells, integrate into them, and go silent. Such infections are called latent: the virus is present in the body but does not actively replicate and does not cause symptoms.
A classic example is the Epstein-Barr virus (EBV), which is found in the vast majority of adults on the planet. Or the herpes virus HHV-7, which nearly everyone catches in childhood. Then there are anelloviruses — they are detected in 80–90% of the population, but scientists still don’t know exactly how they affect health.
All these viruses share one trait: the immune system cannot completely destroy them. It can only contain them, preventing them from replicating. And it is precisely the effectiveness of this containment that determines whether the virus remains a harmless passenger or eventually manifests as disease.
What is viral load in humans
A team of scientists analyzed blood and saliva sample data from more than 917,000 people across three major medical databases. The researchers searched for fragments of viral DNA in biomaterials from healthy individuals — those in whom the infection had not progressed to disease.
For each participant, they calculated what is known as viral load — the amount of viral genetic material circulating in the body. This indicator reveals two things simultaneously: which specific viruses are present and how well the immune system is coping with suppressing them. The higher the viral load, the worse the body is controlling the infection.
The researchers then compared viral load levels with participants’ genetic data to find DNA regions that influence the body’s ability to keep viruses in check.
What is the major histocompatibility complex
The result was impressive: scientists discovered 82 specific loci (regions) in the human genome associated with the level of viral DNA in the body. Particularly many of these regions were found in the so-called major histocompatibility complex (MHC) — a genomic area that can be called the immune system’s command center.
The MHC is responsible for how immune cells distinguish “self” from “non-self.” If certain MHC regions work less effectively, it becomes easier for a virus to hide and avoid destruction. Essentially, an individual’s genetics determine how “comfortable a refuge” their cells will be for latent viruses.
But genetics is not the only factor. The study showed that viral load also depends on age, sex, and even whether a person smokes. Most of the studied viruses turned out to be more prevalent in men compared to women.

The major histocompatibility complex is a key genomic region that controls the immune response to viruses
How one dormant virus triggers different diseases
One of the most interesting discoveries concerns the Epstein-Barr virus. Its viral load, as it turned out, increases with age, rising in winter and declining in summer. The herpes virus HHV-7, on the other hand, begins to decrease roughly from midlife onward.
Using Mendelian randomization (a statistical tool that allows separating cause-and-effect relationships from mere coincidences), the scientists examined how viral load is associated with specific diseases.
It turned out that a high EBV viral load is a direct risk factor for developing Hodgkin’s lymphoma (cancer of the lymphatic system). This is an important finding: it means that antiviral drugs could theoretically reduce the risk of this type of cancer. However, this idea still needs to be verified in clinical trials.
The situation with multiple sclerosis turned out to be more complex. EBV has long been considered one of the triggers of multiple sclerosis, but the link between viral quantity and MS risk was not confirmed in this study. This means that the issue is not how much virus is in the body, but how exactly the immune system responds to it. The immune response, rather than the virus itself, may trigger the autoimmune process.
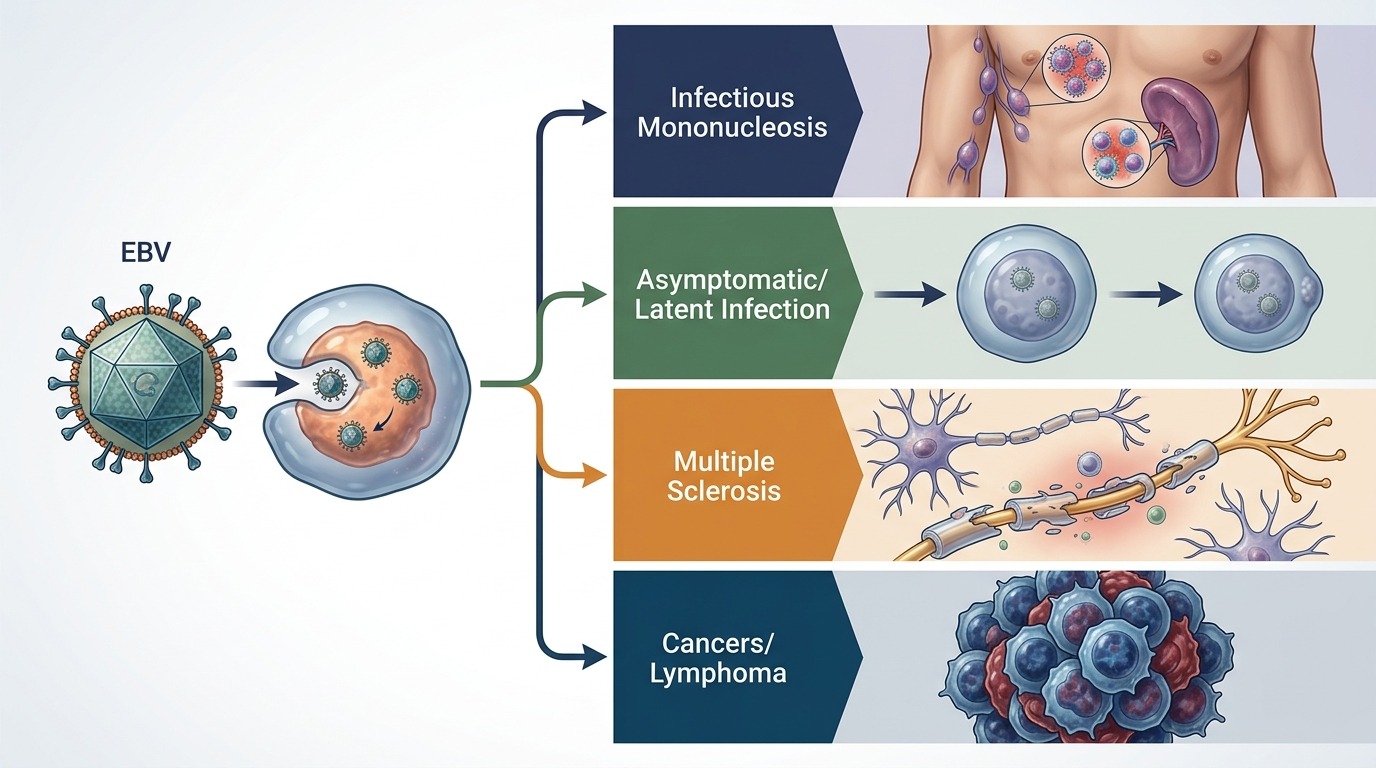
The same Epstein-Barr virus can lead to different outcomes depending on genetics and immune response
What scientists still don’t know about viruses
Despite its scale, the study has an important limitation: the researchers studied only DNA-containing viruses — those that hide within the DNA of cells. RNA viruses, which include coronaviruses, for example, work differently and were not included in this study.
Furthermore, there is another category of viral “residents” in our bodies: ancient viruses that integrated into the human genome thousands and millions of years ago. They have long lost the ability to replicate, but their genetic traces still affect health — sometimes in the most unexpected ways, including influencing predisposition to addictions.
“It’s striking how much DNA can tell us about dynamic biological processes and how our habits, genes, and biology shape those processes,” says geneticist Steven McCarroll, one of the study’s authors.
This research opens the door to personalized medicine for viral infections. If we know which genetic features make a person more vulnerable to a specific latent virus, risks can be assessed in advance — and it may be possible to prevent diseases that these viruses can cause after years of silent presence in the body.


