
Milk starts rising in the pot — a familiar sight for anyone who has ever heated it on the stove
Milk on the stove behaves treacherously: turn away for a minute, and it’s already flooding the burner. Meanwhile, water can be boiled without supervision. Why do two liquids that boil at practically the same temperature behave so differently? The answer lies not in temperature, but in composition. There’s something in milk that makes it escape from the pot.
What Milk Is Made Of
To understand why milk boils over, you first need to figure out how it differs from water. Water is essentially a homogeneous liquid: H₂O molecules and a small amount of dissolved minerals. Milk is far more complex.
Water makes up about 87% of milk, while the remaining 13% consists of proteins, fats, carbohydrates, enzymes, and vitamins. The typical composition of cow’s milk is: water — 86.6%, fat — 4.1%, protein — 3.6%, lactose — 5%.
You might think, 87% water — what difference does it make? But it’s precisely those remaining 13% that cause the “escape.” Proteins and fats are the main culprits: when heated, they behave nothing like water.
At the same time, the boiling point of milk is almost no different from water. It equals 100.2–100.5 degrees. The difference from water is just fractions of a degree. So it’s definitely not because milk “overheats.”
Why Milk Spills Over When Boiling
When milk heats up, it triggers a chain of events that simply doesn’t exist with water. Here’s how it works, step by step.
Stage one — film formation. When heated, the internal structure of milk proteins changes: they become stiffer and begin to clump together with fat droplets. This phenomenon is called coagulation — essentially, proteins and fats form a gel-like mass. Upon contact with air, casein on the milk’s surface partially transitions to a gel-like state as early as 40 degrees, forming a milk skin. Since this mass is lighter than water, it floats to the top and covers the surface of the milk with a thin but fairly strong film.
Stage two — a trap for steam. Meanwhile, at the bottom of the pot, the water in the milk starts turning into steam. Steam bubbles rise upward and hit that very film made of proteins and fats. In regular water, bubbles freely reach the surface and pop. But here — the film won’t let them through.
Stage three — foam and escape. Water vapor pushes against the film from below, stretching it and forming bubbles — much like air inflates a soap bubble. But steam arrives faster and faster, more and more bubbles form, and they build up into a thick layer of foam. The foam grows, rises — and at some point spills over the edge of the pot. There’s your boiled-over milk.
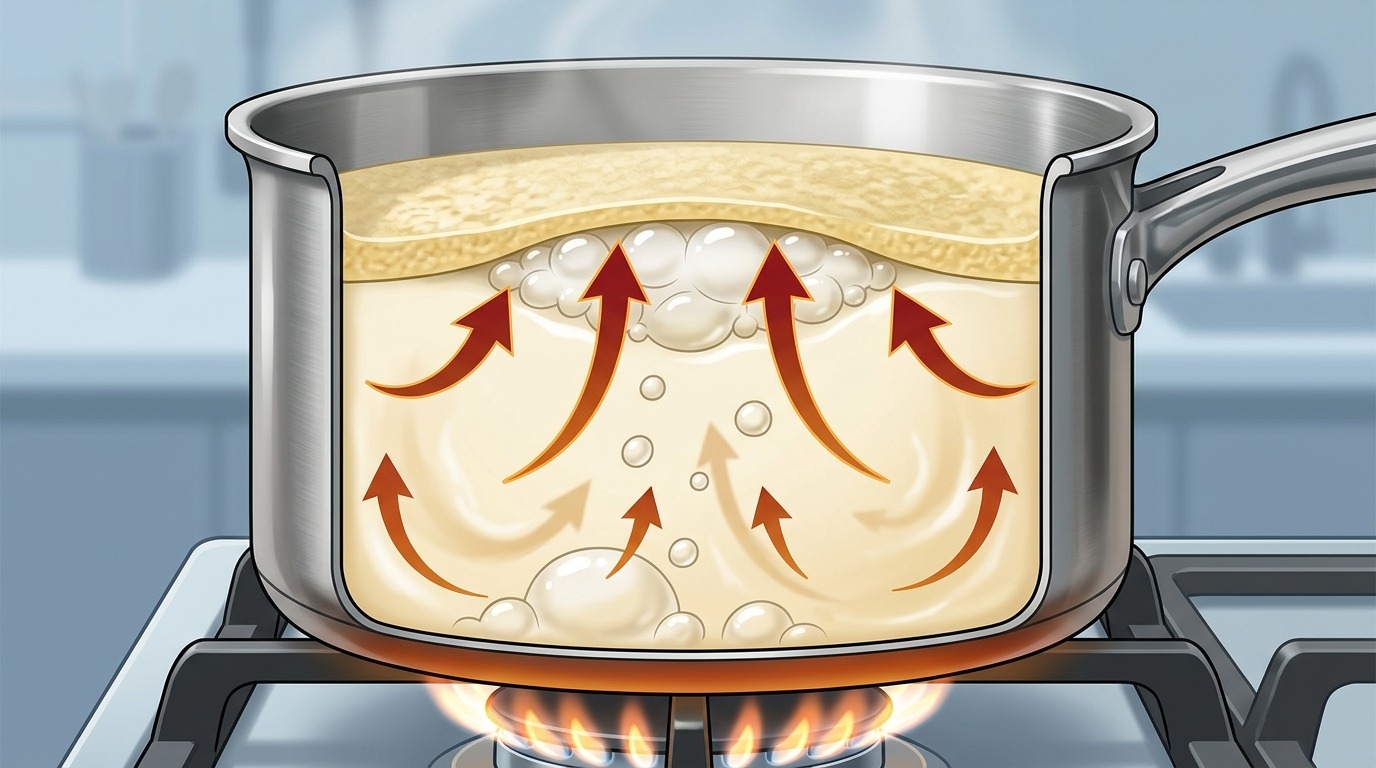
Diagram of what happens inside the pot: steam cannot pass through the film of proteins and fats
Why Water Doesn’t Boil Over Like Milk
Nothing like this happens with water for one simple reason: water contains no proteins or fats that could create a trapping film on the surface. Water vapor bubbles freely rise from the bottom, reach the surface, pop — and the steam escapes into the air. No barrier, no foam, no escape.
You can think of it this way: water is an open door through which steam calmly exits. Milk is a door that has been barricaded with a film of proteins and fats. Steam pushes against the “door” from the inside, it bulges into bubbles, and eventually all the contents spill out.
What to Do to Keep Milk From Boiling Over
Now that we know the mechanism, we can fight each step of it. Here are proven methods:
- Stir the milk — the most reliable option. Stirring breaks the film on the surface and allows steam to escape freely. At the same time, the protein-fat mass is redistributed back into the body of the liquid. But stop stirring, and the film will start forming again.
- Place a spoon or wooden spatula across the pot — this creates a “corridor” for steam to escape. The spoon lifts the film, giving bubbles a way out. However, with strong heating, steam forms faster than it can escape, and the milk may still boil over.
- Use wide cookware — in a large pot or sauté pan, foam bubbles are larger, and the larger the bubble, the less stable it is. At some point, the surface tension of the protein-fat film is no longer enough to hold the bubble intact — and it pops, preventing foam from building up. However, for a small amount of milk, you may need disproportionately large cookware.
There’s also a universal tip: don’t bring milk to a vigorous boil. Heat it on medium and remove it as soon as you see the first bubbles around the edges. For most cooking tasks like porridge, cocoa, or sauce, this is more than enough.


