
Schematic diagram of a lithium-ion battery with a liquid electrolyte that poses a fire hazard
Lithium-ion batteries are the foundation of nearly all modern portable electronics and electric vehicles. And they still use flammable liquid electrolytes. It’s because of these electrolytes that laptops, electric scooters, and electric cars occasionally catch fire. A safe alternative has existed for a long time — polymerized ionic liquids (PIL), which don’t burn. But they had a serious problem: ions moved through them too slowly. A team of researchers figured out why this happens and proposed a solution that increases conductivity up to tenfold. And this could change a lot.
Why Lithium-Ion Batteries Catch Fire
Inside every lithium-ion battery there is an electrolyte — a substance through which lithium ions travel between electrodes during charging and discharging. In most modern batteries, the electrolyte is liquid and contains organic solvents. These solvents conduct ions excellently, but they have a critical drawback: they are highly flammable.
That’s why a damaged or overheated battery can catch fire or even explode. The safety issue with lithium-polymer batteries is especially acute in electric vehicles and devices with fast charging, where currents and temperatures are higher.
Polymerized ionic liquids (PIL) have long been considered as a replacement: they don’t burn and don’t evaporate. But until now they lacked speed — ions moved through them too slowly for practical use.
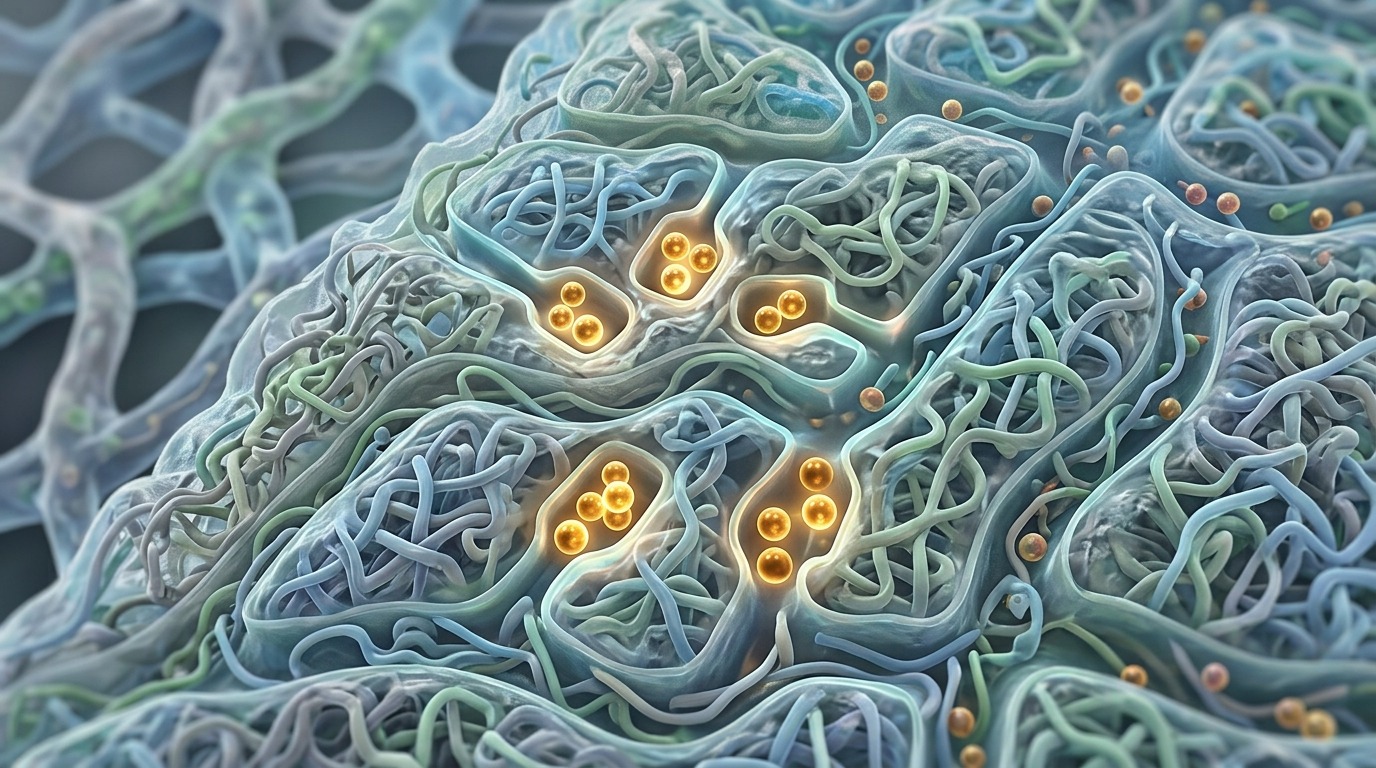
Nanostructure of a polymer with layered organization: dead-end defects trap lithium ions.
How Polymer Electrolytes Work in Batteries
To make PILs strong enough for real devices, researchers combine them with a rigid polymer. The result is a so-called block copolymer — a material in which soft ion-conducting and rigid structural blocks alternate.
These blocks “don’t like” to mix with each other and spontaneously arrange themselves into ordered nanostructures — something like a layered cake at the molecular level. It is along the soft layers of this “cake” that lithium ions are supposed to travel. It sounds elegant, but in practice the structure turned out to be far from perfect.
“Block copolymers are an amazing combination of chemistry and self-assembly,” explains researcher Samuel Adoti. “Even a small change in chemistry can significantly affect how the material organizes and behaves.”
Why Polymer Electrolytes Conducted Ions Poorly
The team’s main discovery, led by Gila Stein, was the identification of “ion traps” inside the polymer. During nanostructure self-assembly, defects inevitably arise — disruptions in the orderliness of the layers. The researchers hypothesized, and then experimentally confirmed, that these defects act as dead ends: ions enter them and get stuck, never reaching the electrode.
“The self-assembly process contains many imperfections,” says Stein. “We hypothesized that some of these defects act as dead ends and block the movement of ions out of the material.”
To test the hypothesis, the team created several versions of the material with different structures and tracked how the changes affected conductivity. The result was unequivocal: the fewer defects — the faster the ions.
How the New Polymer Increased Battery Conductivity 10-Fold
The researchers focused on lamellar (layered) structures because they make it easiest to track ion movement. After studying the relationship between types of defects and conductivity, they developed a set of design rules for creating block copolymers with a minimum number of “dead ends.”
Result: ionic conductivity increased by an order of magnitude — approximately tenfold — while maintaining the mechanical strength of the material.
This is fundamentally important because previously engineers faced a choice: either a soft material that conducts ions well but is too fragile for a real device, or a strong but slow one. In essence, the team showed that the problem was not with the material itself, but with the quality control of its internal architecture.
“This system of alternating layers clearly demonstrates how ionic components affect the spacing between layers, ion mobility, and the structural stability of the block copolymer,” explained Samuel Adoti.

Researcher with a thin polymer film — a potential electrolyte for safe batteries.
How the New Polymer Will Make Batteries Safer and Faster
Increasing ionic conductivity in solid-state electrolytes is one of the key challenges on the path to truly safe and fast-charging batteries. If ions move faster, the battery can charge and deliver energy more efficiently, and the absence of a flammable electrolyte eliminates the main cause of fires.
For now, this is a fundamental study published in the journal Macromolecules. Commercial batteries with such electrolytes are still a long way off: the material needs to be tested in real cells, and durability and production costs need to be evaluated.
However, the design principles developed are applicable not only to batteries. The authors note that the same approaches could be useful in thin-film electronics and actuators — devices that convert electrical signals into mechanical movement.
The main value of the work is not a specific material, but the understanding of why safe polymer electrolytes have until now been inferior to liquid ones. Now that the cause has been found and a way to combat it has been described, developers of next-generation batteries have a clear guideline: not just to make the polymer stronger or softer, but to control order at the nanoscale. And this is possibly exactly the step needed for non-flammable batteries to finally become not only safe but also fast enough.


