
Russia’s cancer vaccine is ready, and the first patient has received it. Image source: gazeta.ru
In Russia, the first patient has received a domestic personalized mRNA cancer vaccine — “Neoonkovak”. The drug was administered to a 60-year-old resident of the Kursk region with skin melanoma. This is not a preventive vaccination in the traditional sense, but a therapeutic vaccine: it does not prevent the disease but helps the body fight an already existing tumor.
How the Russian Cancer Vaccine Works
The word “vaccine” here can be misleading. We are accustomed to vaccines being administered before illness — for flu, measles, or COVID. But “Neoonkovak” is an entirely different story. It is a therapeutic vaccine: it does not prevent disease but helps the body fight an already existing tumor.
The mechanism of action is similar to mRNA vaccine technology used for COVID-19, but instead of a viral protein, the drug encodes unique tumor proteins — neoantigens. In simple terms: every cancer has mutations that distinguish cancer cells from healthy ones. The vaccine “shows” the immune system these mutations so that it learns to recognize and attack them.
The process begins with a biopsy. Then, in a laboratory, genetic analysis of the tissues is performed, mutations are identified, and individual mRNA is synthesized based on them. The finished vaccine is administered to the patient, triggering an immune response against the tumor and metastases. This is exactly why the drug is called personalized — the vaccine is created individually for each patient.
Who Received the First Cancer Vaccine
According to Gazeta.ru, the first person to receive “Neoonkovak” is a 60-year-old resident of the Kursk region diagnosed with skin melanoma. The drug was administered at the National Medical Research Center for Radiology of the Russian Ministry of Health. According to the Ministry of Health, the patient is currently undergoing a course of immunotherapy.
The ministry emphasized that despite the ongoing treatment, the patient still has a high risk of cancer progression. The vaccine is being considered as an additional means of disease control, not as a replacement for the main therapy.
At the initial stage, “Neoonkovak” is planned for use in the most severe cases — in adult patients with inoperable or metastatic skin melanoma, in combination with immunotherapy. The second option is using the vaccine after surgical treatment of melanoma, when metastatic foci have been removed but therapy continues to reduce the risk of recurrence.
Who Developed the Russian Cancer Vaccine
The drug was developed on a messenger RNA (mRNA) platform by specialists from the Gamaleya National Research Center for Epidemiology and Microbiology in collaboration with the Blokhin National Medical Research Center of Oncology and the National Medical Research Center for Radiology of the Russian Ministry of Health. These are three of the largest federal research centers. The Gamaleya Center is also known for developing the Sputnik V COVID vaccine.
The Ministry of Health issued approval for clinical use of the mRNA vaccine “Neoonkovak” in November 2025. The first test batches of the vaccine for quality control were produced in December 2025. Preclinical studies in animals showed that the effectiveness of the domestic vaccine in terms of primary tumor disappearance was 100%, and for metastases — 90%.
Why Melanoma Was Chosen for the Vaccine
The choice of melanoma was not accidental. It is one of the most aggressive types of skin cancer, but it has a feature that makes it a convenient target for immunotherapy. Melanoma cells, especially those caused by ultraviolet exposure, accumulate a huge number of mutations. This means they produce many neoantigens, which are ideal “targets” for vaccine-trained immunity.
The more mutations there are, the more “identification marks” appear on the surface of cancer cells. This makes it easier for the immune system to recognize and destroy them. For comparison: with melanoma, 40–50% of patients are suitable for vaccination, while with prostate cancer — only 5–10%.
Experts point out that mRNA vaccines are applicable “far from all types of tumors, but only to those that have a high antigenic load” — meaning they carry a set of molecular features that easily distinguish them from normal cells.
In the future, the technology is planned to be applied to other types of cancer — lung and kidney. In addition, drugs for the treatment of small cell lung cancer are in development, and in the future — for kidney, breast, and pancreatic cancers.
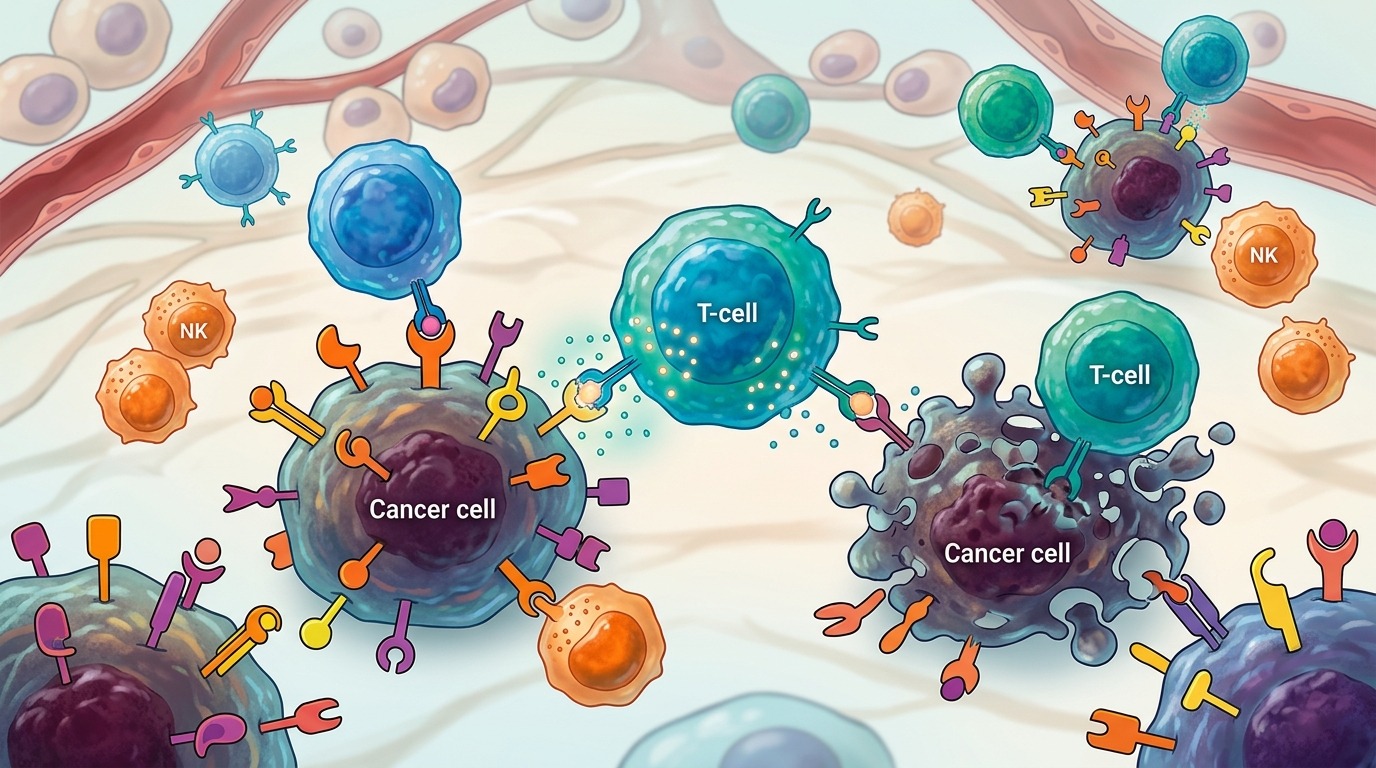
Immune cells attack the tumor by recognizing its mutations
How Effective Is the Russian Cancer Vaccine
Reader skepticism is understandable: promises to “cure cancer” are made regularly, yet the disease still claims millions of lives. But in this case, we are not talking about a miracle or a magic pill. mRNA cancer vaccines are a real and actively developing field in global oncology.
Moderna, BioNTech, as well as dozens of startups and academic centers are working on creating personalized mRNA vaccines designed to help the body recognize and destroy tumor cells. Moderna and Merck are conducting research on an anti-cancer mRNA vaccine that reduces the risk of death and melanoma recurrence. In the second phase of trials, the combination of the vaccine with immunotherapy reduced the risk of recurrence by almost half.
But it is important to honestly state the current status:
- As of today, mRNA vaccines against cancer are still predominantly in phase 1–2 trials;
- There are no confirmed phase 3 results yet; there are no FDA or EMA regulatory approvals for therapeutic mRNA vaccines;
- The Russian “Neoonkovak” has only undergone preclinical trials in animals and is now being administered to the first patients;
- Mass use of the vaccine is not yet on the table. First, its effectiveness and safety must be carefully evaluated in clinical practice. The drug has narrow therapeutic indications and will be available to a limited number of patients.
This is not fiction, but it is not a ready-made medicine for everyone either. This is the initial stage of transitioning from laboratory experiments to real patients — and this is exactly the moment Russia is now marking.
What Cancer Vaccines Exist in Russia
“Neoonkovak” is not the only Russian development. On November 20, 2025, the Russian Ministry of Health issued approvals for clinical use of two individual biotechnological drugs: the mRNA vaccine “Neoonkovak” and the peptide vaccine “Onkopept.” Both will be used in anti-tumor treatment.
The peptide therapeutic cancer vaccine “Onkopept” was created for the treatment of colorectal cancer. Preclinical study results showed high effectiveness and safety of the vaccine. “Onkopept” is designed for people with metastatic colorectal cancer who have already undergone several lines of therapy and need new treatment methods.
In addition, personalized anti-cancer vaccines are planned to be included in the Mandatory Health Insurance (OMS) program: starting in 2026, cancer patients will be able to receive innovative treatment free of charge. If this happens, the cost of treatment for patients will be covered by the state.
All of this represents early stages. None of these drugs has yet proven its effectiveness on a large number of patients in large-scale clinical trials. But the very fact of transitioning from the laboratory to real patients is a step that oncologists have been waiting for years. It is worth following not the loud headlines, but the treatment results of the first patients: they will show whether “Neoonkovak” lives up to the hopes placed on it.


